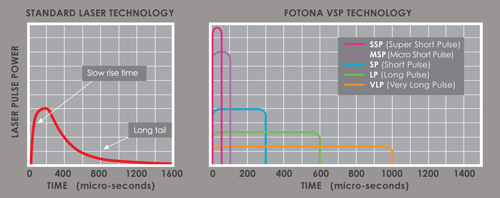
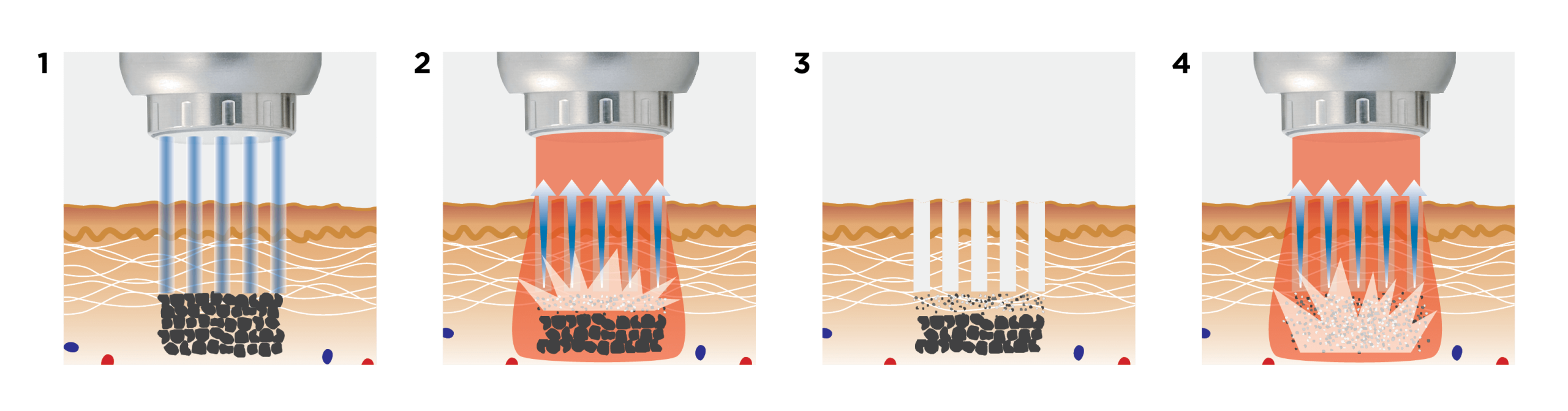
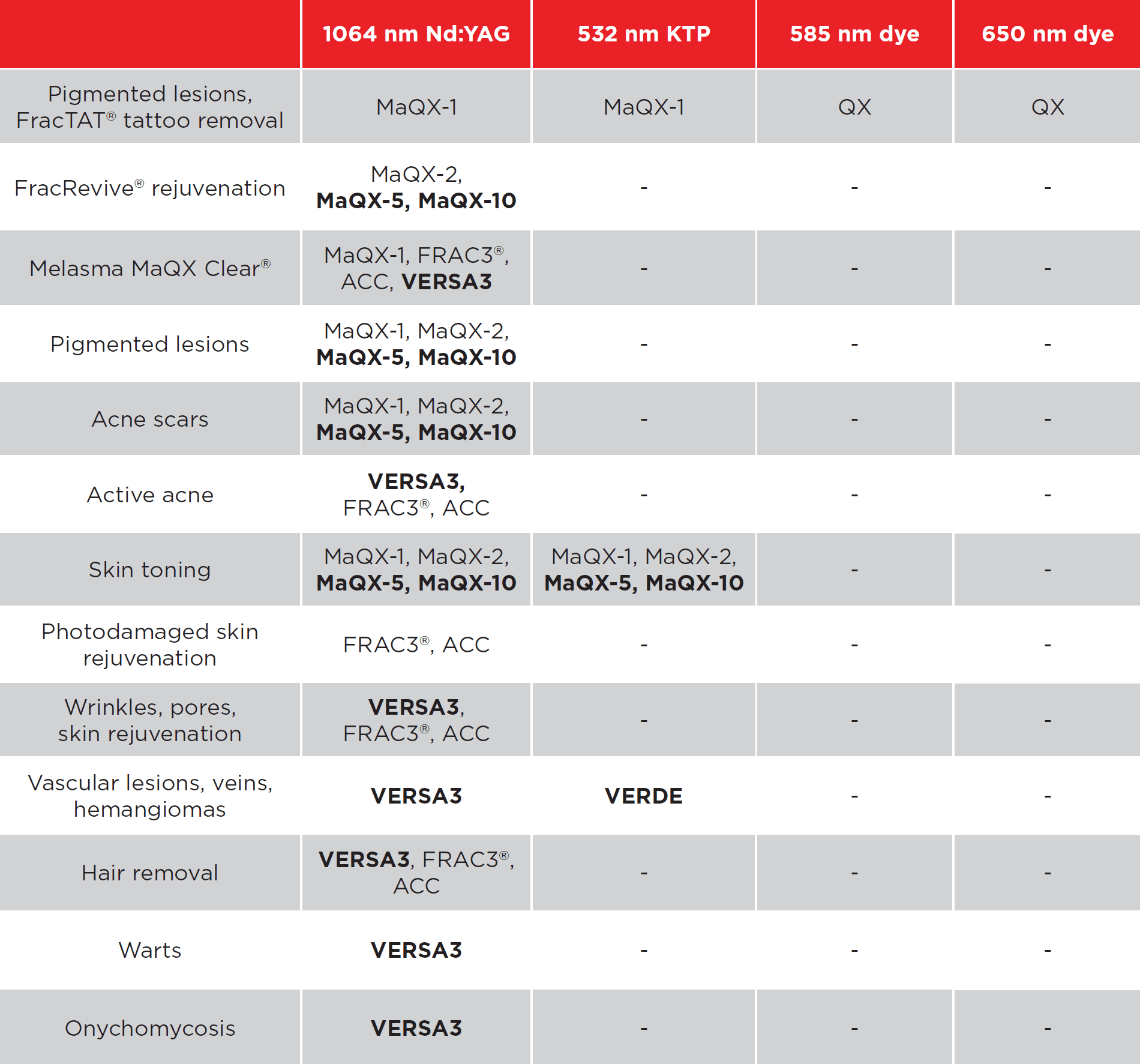
Supplying a medical aesthetic laser to a clinic is not the same as delivering a standard piece of beauty equipment. In the UK, clinics expect more than just a machine. They want a complete supply pathway that covers product legitimacy, training, safety, installation, treatment planning, ongoing support, and commercial confidence. That is especially true with a premium platform like Fotona, which is typically positioned as a long-term clinical investment rather than a simple one-service purchase. Fotona’s own materials emphasise ongoing support, expert training, and multiple applications for each device, which shows that supply in this category is expected to include more than the physical handover of the system.
For clinics across England, Scotland, Wales, and Northern Ireland, the process also sits within a wider regulatory and operational framework. UK medical devices are subject to market and registration rules, and UK guidance makes clear that medical devices placed on the market must follow the relevant legal pathway. For Great Britain, MHRA guidance explains the registration framework and the role of a UK Responsible Person where the manufacturer is based outside the UK, while other guidance sets out the broader UKCA and CE marking picture for products placed on the Great Britain market.
The first expectation: a proper medical device supply route
When a clinic buys or receives a laser system, the first thing it should expect is a clear and legitimate supply route. That means the device should not simply appear through informal channels with vague documentation. In the UK, medical device supply has to sit within the correct regulatory structure. MHRA guidance explains that where a manufacturer is based outside the UK, a UK Responsible Person is required for the registration process in Great Britain, and the MHRA Public Access Registration Database exists so registrations can be checked publicly.
This matters because clinics are becoming more careful about provenance. A serious clinic will want to know who is supplying the device, what support comes with it, whether the product is appropriately registered, and how servicing and documentation will be handled after installation. In practical terms, supplying Fotona lasers into UK clinics usually means being ready to answer these questions early and clearly.
Clinics expect more than delivery alone
Aesthetic clinics do not usually see a platform like Fotona as a plug-and-play purchase. They expect onboarding. Fotona’s own product messaging highlights expert training and ongoing support as part of the value of its systems, and that sets a commercial expectation in the market. A supplier is therefore not only delivering technology. They are helping the clinic prepare to use that technology safely, effectively, and profitably.
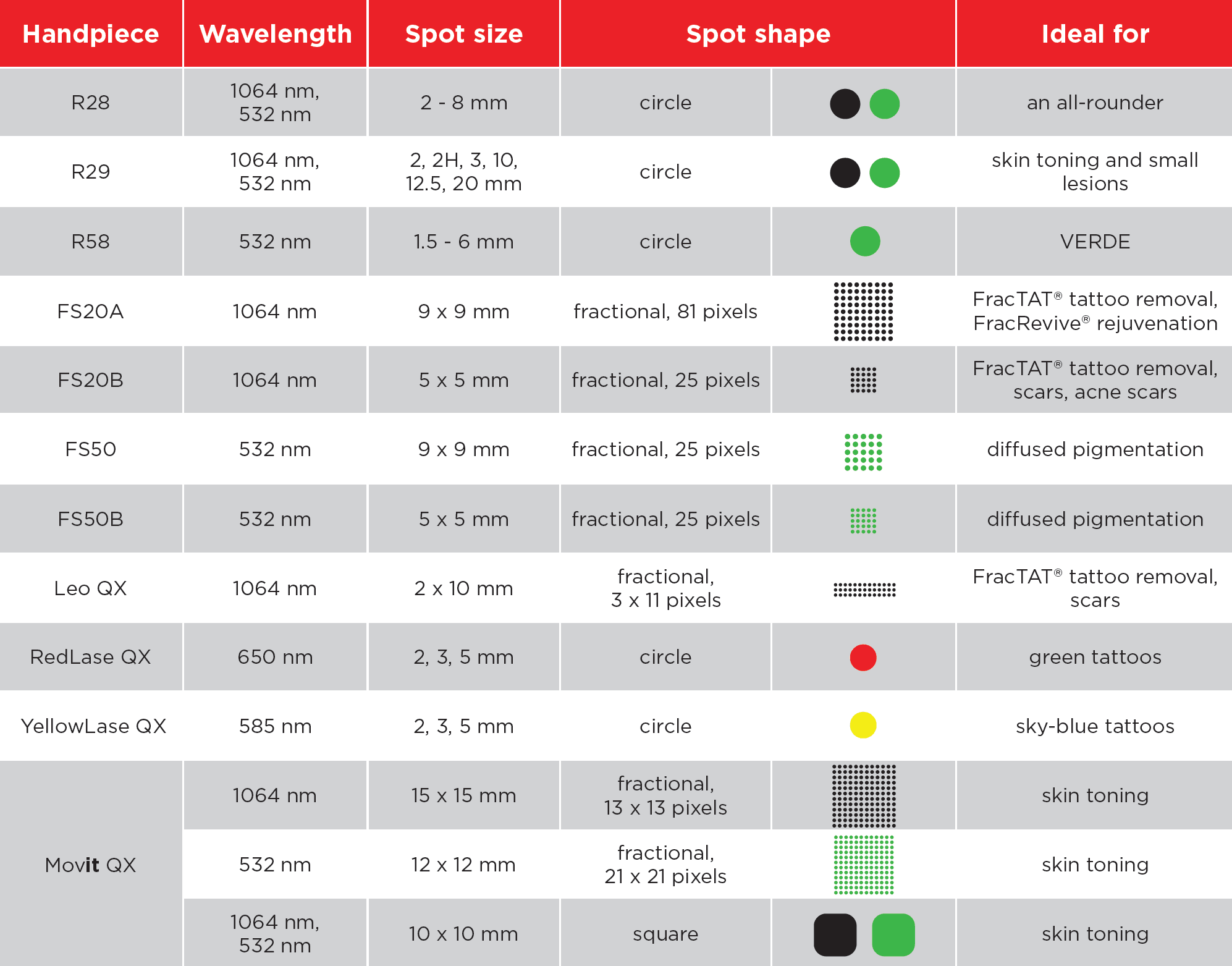
This expectation is even stronger because Fotona is positioned as a multi-application platform. The official aesthetics pages describe a broad treatment range that includes facial rejuvenation, body treatments, resurfacing, tightening, eye-area work, lip treatments, and more. That breadth is commercially attractive, but it also means clinics need help understanding which applications suit their existing patient base, brand position, and practitioner skill level.
Training is part of the supply process
Why clinics expect structured education
Training is one of the most important parts of supplying any medical laser. UK laser practice has a strong safety culture, and the British Medical Laser Association continues to emphasise safety standards, approved training, and best practice across medical and aesthetic laser use. Its website signposts BMLA-approved courses, while its guidance documents cover treatment protocols, record keeping, consent, and technical requirements.
For a clinic, this means the supplier should be prepared to support not only treatment onboarding but also the wider education picture. BMLA material also points to the importance of laser safety roles and awareness training, including Laser Protection Supervisor education and safety-awareness training for staff present during laser use.
Training is also a commercial issue
From a business point of view, training affects more than compliance. It shapes confidence. A practitioner who understands the platform well is more likely to consult properly, recommend suitable treatment plans, and use the device regularly. A poorly supported clinic may own a strong platform but fail to build momentum because staff are hesitant or undertrained. That is why supplying Fotona to UK clinics usually needs to include a realistic conversation about onboarding, team capability, and the time needed to build treatment confidence. Fotona’s own messaging around ongoing support reinforces this expectation.
Safety, governance, and clinic setup
Another thing UK clinics should expect is a strong focus on safety and governance. BMLA guidance for treatment use and independent clinic advice makes clear that laser settings require proper oversight, record keeping, advice structures, and safe operational standards. That means supply is not only about where the machine goes in the room. It is about how the clinic will run it responsibly once it is there.
This is particularly relevant because regulatory attention around non-surgical cosmetic procedures continues to evolve. The UK government’s consultation on licensing non-surgical cosmetic procedures in England underlines the direction of travel: greater emphasis on standards, patient confidence, and safe delivery. Even where rules are still developing, reputable clinics and suppliers are already expected to behave as though scrutiny matters.
Commercial expectations: clinics want a platform that can earn
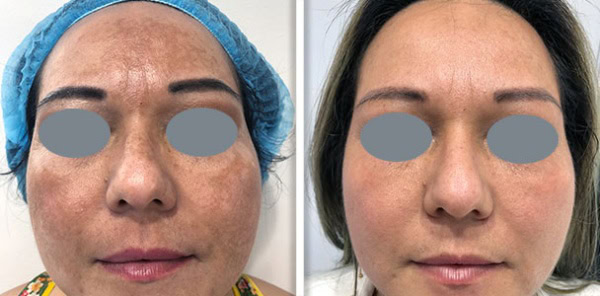
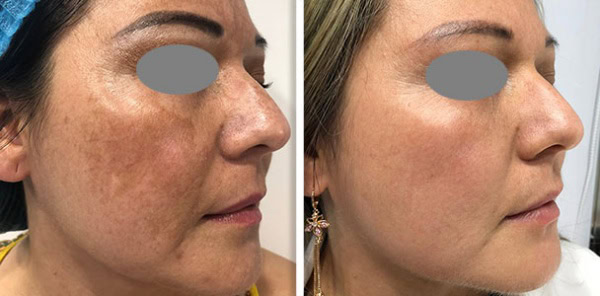
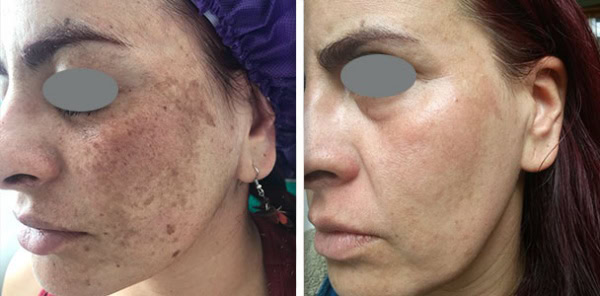
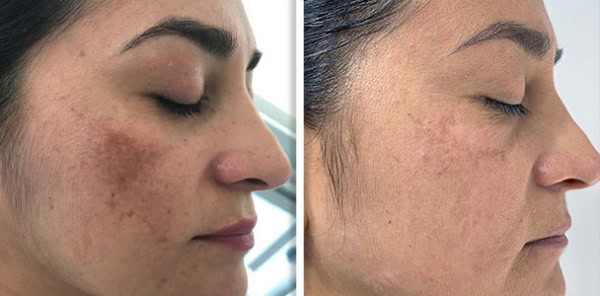
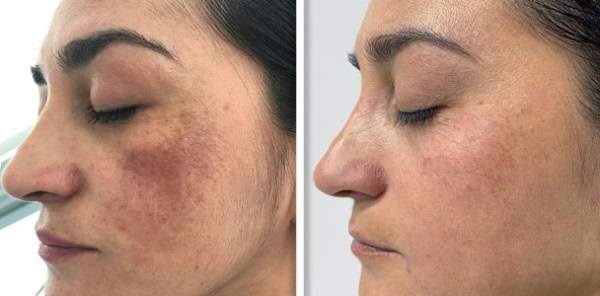
Supplying a Fotona system to a clinic is never only a clinical conversation. It is also a commercial one. Clinics want to understand whether the device will generate enough demand and revenue to justify the investment. Public UK clinic pricing gives a useful indication of how Fotona treatments are currently positioned in the market. One UK clinic lists premium pricing for Fotona-led services, including Fotona 4D face lift, SmoothEye, TightSculpting, NightLase, and LipLase, showing that these services are often sold as high-value treatments rather than low-ticket add-ons.
That matters when supplying across the UK because clinic buyers are usually asking the same practical questions. Will this platform help us compete? Can we use it across multiple services? Will patients understand the value? Can it support a premium consultation model? These are sensible questions, especially in a market where clinics are trying to avoid buying separate devices for every new treatment trend.
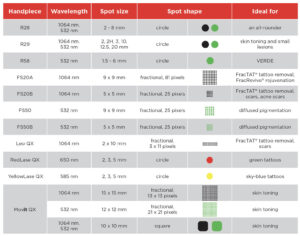
Fotona services
Fotona services are attractive to UK clinics because they support a broad treatment menu from one recognised platform. Official Fotona materials position the brand across facial rejuvenation, tightening, resurfacing, body applications, and other aesthetic uses, while UK clinics already market services such as Fotona4D, SmoothEye, TightSculpting, NightLase, and LipLase at premium price points. In practical terms, this gives clinics the chance to build more personalised treatment plans and create more than one revenue stream from the same technology.
Support after installation is part of the deal
A clinic should also expect supply to continue after installation. Fotona’s own US-facing product messaging explicitly highlights ongoing support and expert training, and that matters because no clinic wants to invest in a laser platform only to be left alone once the device is delivered. Whether the questions are technical, commercial, or training-related, suppliers in this category are expected to remain involved.
This is especially true for multi-platform systems, because the real value is often unlocked gradually. A clinic might begin with one or two core treatments, then expand into more applications over time. That kind of growth is easier when the supplier relationship includes follow-up support, refresher education, and guidance on how to introduce additional protocols.
What clinics across the UK should realistically expect
In simple terms, a UK clinic buying or receiving a Fotona laser should expect five things. First, a proper legal and regulatory supply route. Second, clear onboarding and training. Third, support safety and governance expectations. Fourth, honest commercial guidance on how the system can fit the clinic’s model. Fifth, ongoing support after installation rather than a one-time transaction. Those expectations are grounded in current UK device guidance, BMLA safety standards, and Fotona’s own brand positioning around training, support, and multi-application use.
Final thoughts
Supplying Fotona lasers to clinics across the UK is about much more than equipment logistics. It sits at the intersection of regulation, safety, training, business planning, and after-sales support. Clinics want a supplier who understands all of those layers because the machine itself is only one part of the investment. The most successful supply relationships are usually the ones where the clinic feels supported from the first conversation through to installation, training, and the first months of real patient use. That is what UK clinics increasingly expect, and with a premium platform like Fotona, that expectation is entirely reasonable.
FAQs
What should a UK clinic check before buying a Fotona laser?
A clinic should check the legitimacy of the supply route, the registration and regulatory pathway, the support structure, the training offered, and what happens after installation. MHRA guidance and the public registration database are useful reference points when checking the device pathway in the UK.
Do Fotona lasers come with training and support?
Fotona’s official materials emphasise ongoing support and expert training, which suggests clinics should expect more than just delivery when investing in one of its systems.
Are UK clinics expected to follow laser safety guidance?
Yes. BMLA resources set out treatment guidelines, safety standards, and approved training routes for lasers and IPL devices used in clinical and aesthetic settings.
Which Fotona treatments are most commonly marketed by UK clinics?
Public UK clinic listings show services such as Fotona4D, SmoothEye, TightSculpting, NightLase, and LipLase being actively marketed.
Will regulation affect how aesthetic lasers are supplied in the UK?
Yes. UK medical device regulation already applies to devices placed on the market, and wider policy attention on non-surgical cosmetic procedures shows that standards and oversight remain important.
Why do clinics prefer multi-platform systems like Fotona?
Because they can support multiple treatment categories from one platform, which may improve flexibility, service range, and commercial potential. Fotona’s official pages highlight multiple applications across aesthetics and broader medical use.